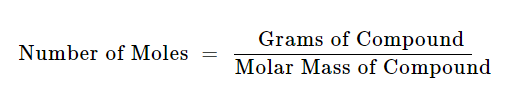
Density is defined as mass per unit volume. The calculator can use any two of the values to calculate the third. The Density Calculator uses the formula p=m/V, or density (p) is equal to mass (m) divided by volume (V). What is the formula to calculate density? Number of moles of S in 1.8 x 1024 S atoms a) 1.0 mole S atoms b) 3.0 mole S atoms c) 1.1 x 1048 mole S atoms. Divide the number of moles of solute by the number of liters of solution.Calculate the number of liters of solution present.So, we need to convert grams of BaCl 2 to moles of BaCl 2. Step 3: Make sure your given is in moles If grams/liters/particles of A, convert the moles of A. 
Calculate the number of moles of solute present. Step 2: Set up the multiplication grid Put the given on the ‘front ledge’, and what you want to find behind the grid.Herein, how do you find moles of NaOH from mL? Similarly, how many moles are in a liter? The equality of 1 mole = 22.4 L is the basis for the conversion factor. 1 mole is equal to 1 moles In, or 114.818 grams. 
Then in the left-hand column, choose Moles. That’s why you can use this conversion calculator to convert from grams to moles How to use the grams to mole calculator In the right column, enter the number of grams. One mole equals one gram per mole (a quantity). You can view more details on each measurement unit: molecular weight of In or grams The SI base unit for amount of substance is the mole. Grams measure weight, while moles measure the number of particles or molecules. We assume you are converting between moles In and gram. Moreover, this is also Avogadro’s number. This, however, scientists have found experimentally. So, 1 mole by that calculation number of atoms that are present in 12 g of C-12 6.022 x 1023 atoms. Moles (mol) Molarity (M) x Volume (L) 0.5 x 2 1 mol For. Now, the amount of mass that you find in 1 mole is the same as the number of atoms in exactly 12 grams of pure carbon-12. To convert formula units to moles you divide the quantity of formula units by Avogadro's number, which is 6.022 x 1023 formula units/ mole.įurthermore, how many moles are in a gram? First you must calculate the number of moles in this solution, by rearranging the equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
